Education
- North Dakota State University, Fargo, North Dakota: B.S., 1983.
- University of Illinois Urbana-Champaign, Urbana, Illinois: Ph. D, 1988 (Thesis advisor: Gary B, Schuster).
- University of California, Berkeley, California Postdoctoral Associate 1989 (Postdoctoral advisor: John E. Hearst).
Professional Experience
- Assistant Professor, University of Maryland (1989-1995)
- Associate Professor, University of Maryland (1995-1999)
- Professor, University of Maryland (1999-present)
- Associate Chair for Facilities and Instruction, University of Maryland (2005-2025)
- Chair Department of Chemistry & Biochemistry (2025-present)
Research Interests
Organic Chemistry, Photochemistry, Physical Organic Chemistry.
The Falvey group studies the mechanisms of photochemical reactions and applies that knowledge to the design of useful systems for applications drug delivery and the design of novel photoresponsive materials. Photochemical methods are also used to generate and characterize unstable molecular species such as nitrenium ions, diradicals and carbocations.
Major Recognitions and Honors
16th Annual Sara Jane Rhoads and Rebecca Raulins Distinguished Lecturer, University of Wyoming. 2008.
- Robert Rowan Memorial Award for Departmental Service- Dept. of Chemistry and Biochemistry University of Maryland 2014.
- Phillip Merrill Presidential Scholar Program Faculty Mentor, University of Maryland 2013 and 2008.
- INNOLEC Visiting Lecturer, Masaryk University, Brno, Czech Republic. October 2-8 2011.
- Merck Award, Department of Chemistry, North Dakota State University, 1983.
- Albert C. Bean Memorial Coatings Scholarship, Department of Polymers and Coatings, North Dakota State University, 1982-1983.
Significant Professional Service and Activities
1. Editorial Advisory Board, Journal of Physical Organic Chemistry. 2004-present
2. International Chemistry Olympiad Scientific Advisory Committee 2012, College Park, MD.
3. Co-Chair, Gordon Research Conference on Photochemistry July 2017, Lewiston, ME.
5. Chair, University Senate, University of Maryland, 2017-2018.
Research Students Mentored
Over 27 undergraduates and 21 Ph. D. students have done research with the Falvey group, most of whom have continued their scientific careers in academia, industry, or government.
One area of research in the Falvey Group is the mechanistic design, synthesis and study of photoreleasable protecting groups. The overall goal is to create a suite of protecting groups whose release can be triggered using inexpensive visible light sources. One specific strategy has been to photoinduced electron transfer from visible-light absorbing dyes to a protecting group that can be activated through photoinduced single electron transfer. Specifically the release of carboxylic acids phenacyl and 4-N-alkylpicolinium esters have been examined. An example of one such reaction is shown below.
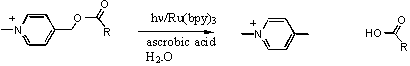
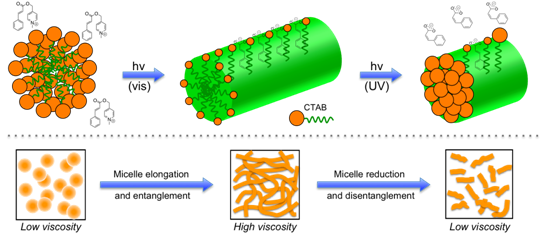
These reactions have recently been applied to the development of phototriggering reagents that can rapidly and reversibly alter the viscosities of bulk liquids by catalyzing self-assembly processes. For example, visible light triggered photorelease of cinnamate ions in the presence of CTAB micelles has been shown to convert the latter from spherical structures into elongated worm-like structures. This in turn causes the solution to convert from a free flowing fluid to a gel-like state.